What are post-translational modifications?
Post-translational modifications (PTMs) involve the covalent addition of functional groups or proteins, proteolytic cleave of regulatory subunits or degradation of entire proteins. [1] Some examples of PTMs include phosphorylation, glycosylation, methylation, acetylation and proteolysis. Post-Translational modifications have significant impacts on various aspects of cell biology and pathogenesis. [1] Studying PTMs are critical to studying cell biology in addition to disease treatment and prevention.
What is protein phosphorylation?
Protein phosphorylation, specifically on serine, threonine or tyrosine residues is one of the most well-studied post-translational modifications. [2] Phosphorylation allows for the regulation of many cellular processes such as cell cycle, growth, apoptosis and signal transduction processes. Although phosphorylation occurs in bacterial proteins, it has a greater presence in eukaryotic cells. Due to the importance of protein phosphorylation, phosphoproteomics has been established as a field of proteomics that focuses specifically on the study of phosphorylated proteins. [2]
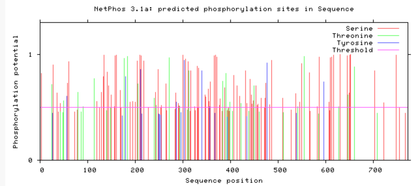
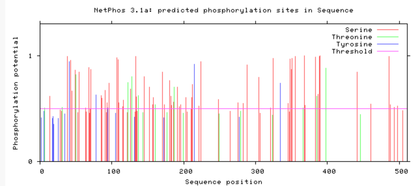
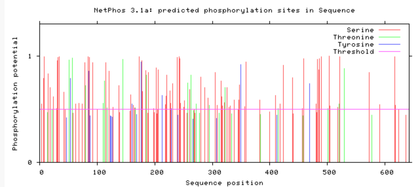
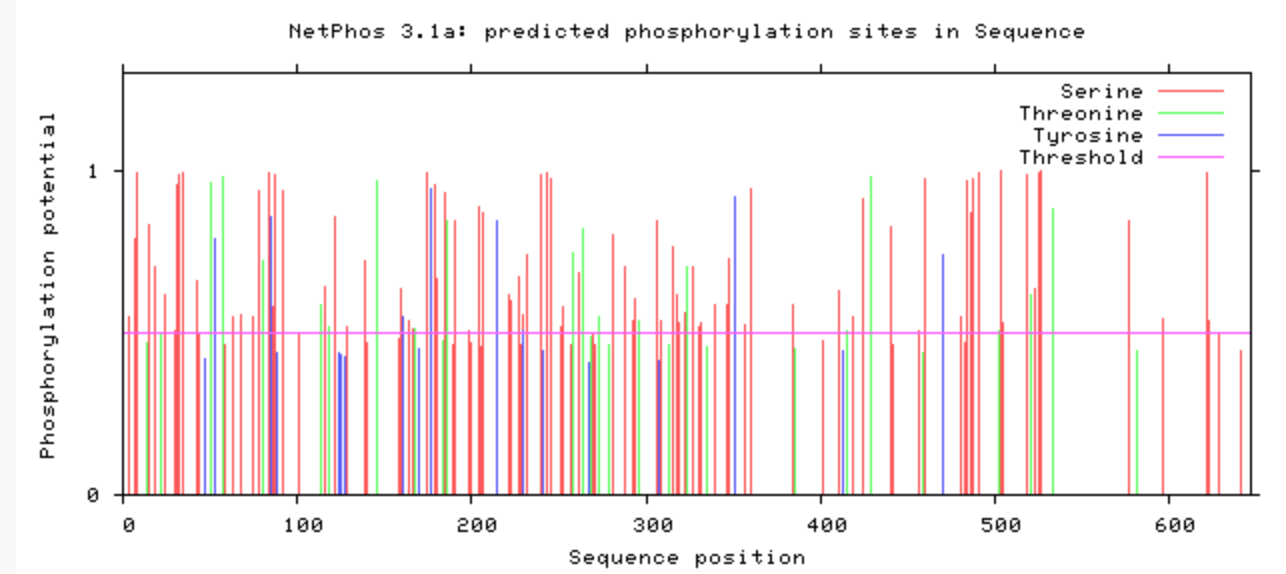
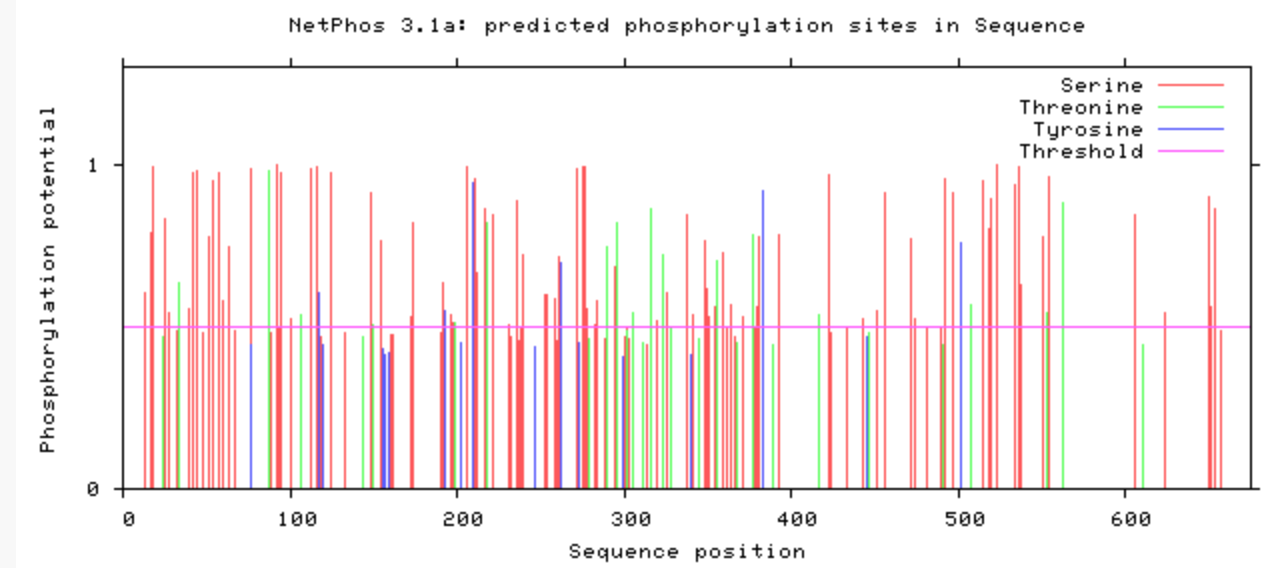
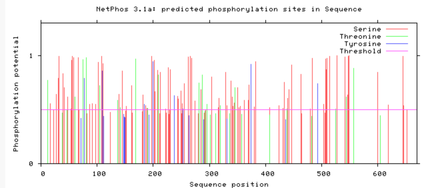
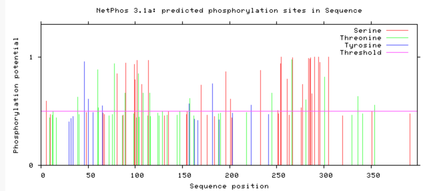
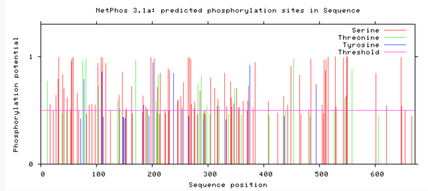
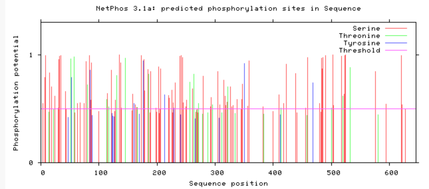
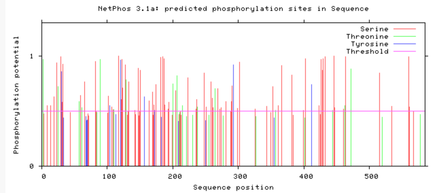
Are the predicted phosphorylation sites similar
across the model organisms?
Conclusion
As seen above, there are many predicted phosphorylation sites for the TCF4 protein. However, it is not certain whether these sites are actually phosphorylated or not. The figures above show that the phosphorylation sites appear to be conserved across the majority of model organisms. D. melanogaster and C. elegans have the most visible discrepancies compared to the phosphorylation sites of other model organisms.
References
Header: http://www.riverrundesign.com/art-of-the-day-dna-interchange-2/
[1] Overview of Post-Translational Modifications (PTMs). (n.d.). Retrieved March 22, 2018, from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[2] Phosphorylation. (n.d.). Retrieved March 22, 2018, from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/phosphorylation.html
Header: http://www.riverrundesign.com/art-of-the-day-dna-interchange-2/
[1] Overview of Post-Translational Modifications (PTMs). (n.d.). Retrieved March 22, 2018, from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[2] Phosphorylation. (n.d.). Retrieved March 22, 2018, from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/phosphorylation.html
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.